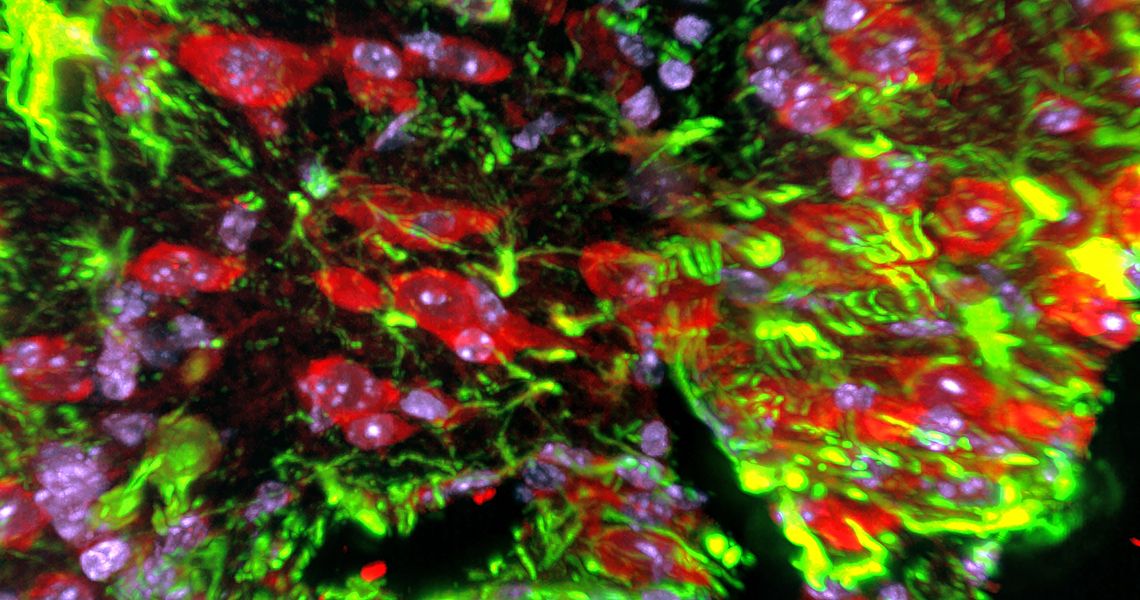
WASHINGTON, D.C. (Dec. 11, 2023) — A new study published in Proceedings of the National Academy of Sciences Nexus provides a better understanding of how the brain responds to injuries. Researchers at the George Washington University (GW) School of Medicine and Health Sciences (SMHS) discovered that a protein called Snail plays a key role in coordinating the response of brain cells after an injury.
The study shows that after an injury to the central nervous system (CNS) a group of localized cells start to produce Snail, a transcription factor or protein that has been implicated in the repair process. The GW researchers show that changing how much Snail is produced can significantly affect whether the injury starts to heal efficiently or whether there is additional damage.
"Our findings reveal the intricate ways the brain responds to injuries,” said senior author Robert Miller, PhD, the Vivian Gill Distinguished Research Professor and vice dean of the GW SMHS. ”Snail appears to be a key player in coordinating these responses, opening up promising possibilities for treatments that can minimize damage and enhance recovery from neurological injuries."
Key findings:
- This study identifies for the first time a special group of microglial-like cells that produce Snail. Microglial cells are found in the central nervous system.
- Lowering the amount of Snail produced after an injury results in inflammation and increased cell death. During this process the injury gets worse not better and there are fewer connections or synapses between brain cells.
- In contrast, when Snail levels are increased the outcome of brain injury improves–suggesting this protein can help limit the spread of injury-induced damage.
The research raises questions about whether an experimental drug that affects Snail production could be used to limit the damage incurred after someone suffers a stroke or has been injured in an accident, Miller said. Additional studies must be done to show that increasing Snail production could curtail injury or even promote healing of the brain.
Miller and his team also plan to study the regulation of Snail in diseases like multiple sclerosis. Multiple sclerosis is a disease resulting in damage to myelin, the protective layer insulating nerve fibers in the brain. If drugs targeting Snail could be used to stop that damage, many of the future symptoms of this disease could be eased, he says.
But researchers have years of work to do before new drugs targeting Snail can be tested in clinical trials. The payoff ultimately might be drugs that can lead to accelerated healing for stroke damage, head wounds and even neurodegenerative diseases like dementia.
In addition to Miller and a team of researchers at the GW School of Medicine and Health Sciences, Cheryl Clarkson-Paredes, PhD, DMD, a senior research scientist at the GW Nanofabrication and Imaging Center, served as the lead author of the paper, “A unique cell population expressing the Epithelial-Mesenchymal Transition-transcription factor Snail moderates microglial and astrocyte injury responses,”
The research was supported by Biogen and the National Institutes of Health.