As the virulent omicron variant sparks a new surge of the coronavirus cases around the world, a key component of reducing transmissions and keeping society open is faster and more accurate testing. Researchers at the George Washington University School of Medicine and Health Sciences (SMHS) are studying a technique that identifies biomarkers of infection in the bloodstream that could produce a new and highly accurate test for numerous diseases and disorders, including COVID-19.
“It’s a bit like inventing a new type of stethoscope,” said lead investigator Tim McCaffrey, PhD, professor of medicine and of microbiology, immunology, and tropical medicine at SMHS. “If we could identify a biomarker to tell us whether the problem is infectious, traumatic or inflammatory, doctors would have a much better idea of how to proceed.”
The interdisciplinary research team includes students from the School of Engineering and Applied Science (SEAS) and collaborators in the SMHS Department of Emergency Medicine.
Their work attracted the interest of Ulvi M. Kasimov and Reykhan Kasimova, noted entrepreneurs and philanthropists. Helping stem the spread of COVID-19 by supporting bench-to-bedside medical research and better detection tools is a priority for the couple.
Inspired by McCaffrey’s progress, the Kasimovs made a $1 million current-use gift in support of the COVID-19 Infection Diagnosis Research Project, with the goal of speeding up development of the diagnostic point-of-care testing device.
“As GW parents who live outside the United States, we are well aware of the devastating impact of the pandemic worldwide, and we were seeking to support research with the potential to improve lives,” said Kasimov. “We are intrigued by Dr. McCaffrey’s innovative and exciting approach to develop more accurate COVID-19 testing and wanted to help make these diagnostics available as soon as possible.”
Gifts of this nature, McCaffrey said, can have an immediate real-world impact on the research timeline. “This rapid infusion of resources,” he said, “takes years off the development process.”
Donna Arbide, GW's vice president for development and alumni relations, said the university is grateful for the Kasimovs’ investment, which “is an incredible example of the impact philanthropy can have on our world.”
“GW is at the forefront of efforts to combat COVID-19, and this extremely generous gift from the Kasimov family will provide substantial support for critical research that could transform lives by transforming health care,” Arbide said.
“As we welcome the Kasimovs to our GW community, we are pleased to extend membership into our university’s two most prestigious donor societies, the 1821 Benefactors of the George Washington Society and the top level of GW’s Family Leadership Circle," she added.
The 1821 Benefactors honor the university’s most generous supporters, and the Family Leadership Circle provides opportunities to engage with university leadership and network with other GW families interested in changing lives through philanthropy.
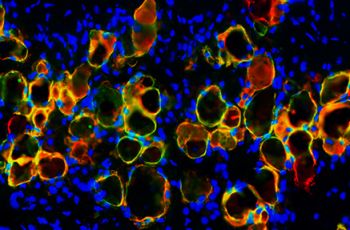
McCaffrey and his research team, including GW SMHS emergency medicine physicians John LaFleur, MD, and David Yamane, MD, and bioengineerings students from GW’s School of Engineering and Applied Sciences conducted a deep analysis of hospitalized COVID-19 patients, including a full series of RNA tests to understand the effect on the human immune system. The team profiled the RNA molecules expressed in the bloodstream to identify any proteins that might indicate an infection. They determined that the severity of COVID-19 infection is associated with significant changes in the activation of neutrophils, a type of protein peptide used by the immune system. They found that the presence of COVID-19 and the severity of the infection were associated with up to a “25-fold increased expression of neutrophils.”
“We could see visible differences in the purified neutrophils cells of people [infected with COVID-19],” he said.
Biomarkers of infection could have numerous uses, according to McCaffrey, particularly by distinguishing infectious diseases from aseptic inflammation, such as that occurring in asthma, allergies, and fibrotic or autoimmune disorders.
McCaffrey recently published a study using a similar RNA expression analysis technique to identify biomarkers for coronary artery disease (CAD). Current CAD diagnostics use imaging equipment to measure restricted blood flow in the heart’s major arteries. Those tests, however, frequently yield false negative results. Results of that study suggest that autoimmunity may play a significant factor in CAD.
“Based on the gene expression pattern in our studies, it appears as though the patient’s immune system becomes stressed, which causes an inappropriate attack on their own arteries,” McCaffrey explained, adding that the immune connection extends to COVID-19 as well. The method, he added, “could transform health care.”